Deliver more. Stress less.
B cells don't get nearly enough love. They present antigen, secrete antibodies, establish long-lived memory, and are quietly powering some of the most exciting therapeutic approaches on the horizon. And yet, when it comes to actually engineering them? Most researchers know the frustration all too well.
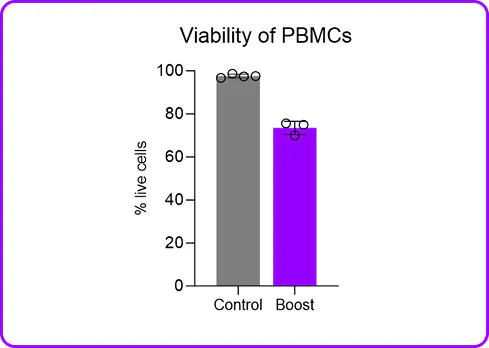
B cells are stubbornly resistant to lentiviral transduction, with standard protocols limping along at low single-digit efficiency. Electroporation can force cargo in, but it torches viability, scrambles gene expression, and leaves you with cells that barely resemble what you started with. Not ideal when your whole experiment depends on healthy, functional B cells.
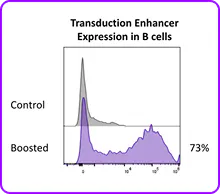
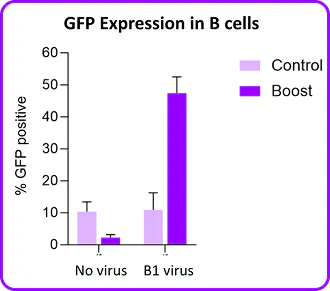
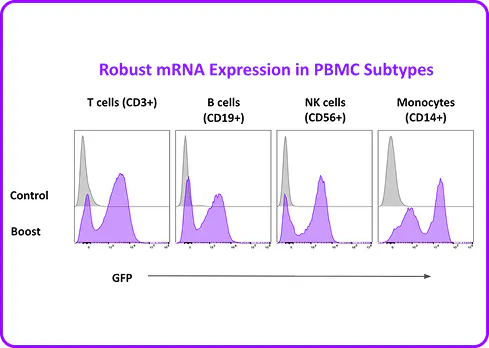
Portal's Gateway system delivers mRNA, proteins, and other cargo directly to the cytosol of primary human B cells through rapid mechanical membrane disruption. No viral vectors, no electrical fields, no apology tour for your cell viability numbers. In mixed PBMC populations, mechanoporation achieves functional mRNA expression in CD19+ B cells alongside T cells, NK cells, and monocytes in a single processing step at high viability. No pre-sorting required. And for researchers who are committed to lentiviral workflows, mechanoporation delivery of a transduction enhancer RNA boosted B cell transduction. It plays nicely with others too.
Below, we break down Portal's B cell delivery data across multiple cargo types and workflows. Your B cells have been through enough. Let's make it easier.